Territorial Availability: Available through Bertin Technologies only in France
- Synonyms
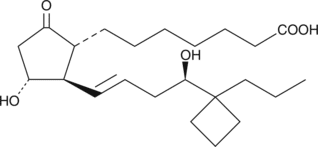
- 9-oxo-11?,16R-dihydroxy-17-cyclobutyl-prost-13E-en-1-oic acid
- Correlated keywords
- PGE2 prostaglandins 5,6-dehydro-butaprost free acids EP2 receptors binding selectivity carboxylic acid selective analogs C-16 (R)-epimers
- Product Overview:
Butaprost is a structural analog of prostaglandin E2 (PGE2) with good selectivity for the EP2 receptor subtype. Butaprost has frequently been used to pharmacologically define the EP receptor expression profile of various human and animal tissues and cells.{3317} Serious confusion as to the structure of butaprost was generated by Gardiner in 1986,{3326} when he reported that the epimer of butaprost showing this selective activity was the C-16 (R)-epimer (See reference 2 and NOTE). In order to increase the binding affinity of (R)-butaprost for prostanoid receptors, we removed the methyl ester of (R)-butaprost and re-established the natural C-1 carboxylic acid. Prostaglandin free acids generally bind to their cognate receptors with 10 to 100 times the affinity of the corresponding ester derivative. The pharmacology of (R)-butaprost has not been carefully studied, but it is generally considered to be the less active C-16 epimer.{3178} (NOTE: In the Gardiner paper in the 1986 British Journal of Pharmacology, butaprost appears on page 46 where it is given the name TR 4979. The structure as drawn is incorrect, in that the author was using and referring to the more active C-16 epimer, which is actually 16(S). The structure on page 46 shows the structure as 16(R). It was not until the late 1990’s that careful studies both in the US and Japan correctly identified the actual configuration of C-16 in the compound called butaprost is 16(S).){3326}
Cayman Chemical’s mission is to help make research possible by supplying scientists worldwide with the basic research tools necessary for advancing human and animal health. Our utmost commitment to healthcare researchers is to offer the highest quality products with an affordable pricing policy.
Our scientists are experts in the synthesis, purification, and characterization of biochemicals ranging from small drug-like heterocycles to complex biolipids, fatty acids, and many others. We are also highly skilled in all aspects of assay and antibody development, protein expression, crystallization, and structure determination.
Over the past thirty years, Cayman developed a deep knowledge base in lipid biochemistry, including research involving the arachidonic acid cascade, inositol phosphates, and cannabinoids. This knowledge enabled the production of reagents of exceptional quality for cancer, oxidative injury, epigenetics, neuroscience, inflammation, metabolism, and many additional lines of research.
Our organic and analytical chemists specialize in the rapid development of manufacturing processes and analytical methods to carry out clinical and commercial GMP-API production. Pre-clinical drug discovery efforts are currently underway in the areas of bone restoration and repair, muscular dystrophy, oncology, and inflammation. A separate group of Ph.D.-level scientists are dedicated to offering Hit-to-Lead Discovery and Profiling Services for epigenetic targets. Our knowledgeable chemists can be contracted to perform complete sample analysis for analytes measured by the majority of our assays. We also offer a wide range of analytical services using LC-MS/MS, HPLC, GC, and many other techniques.
Accreditations
ISO/IEC 17025:2005
ISO Guide 34:2009
Cayman is a leader in the field of emerging drugs of abuse, providing high-purity Schedule I-V Controlled Substances to federally-licensed laboratories and qualified academic research institutions for forensic analyses. We are certified by ACLASS Accreditation Services with dual accreditation to ISO/IEC 17025:2005 and ISO Guide 34:2009.